Newsletter Edition #158 [Treaty Talks]
Readers,
Making decisions based on incomplete information is a quotidian reality for policymakers. But it is totally unwholesome to those tasked with writing up laws.
This is the predicament that health negotiators are facing in the discussions on the Pathogen Access and Benefit Sharing system at the WHO. Countries do not have an estimate of the size of a future market for a pathogen with pandemic potential, but many are of the view that clear contractual obligations from manufacturers of medical products will be important. This they believe, will ensure a commitment to equity, and on a more practical level, to push through ratification in Parliaments.
In our sponsored guest edition today, legal expert Adam Strobeyko at the Geneva Graduate Institute's Global Health Centre, lays out an analysis on what could work in this twilight zone of the negotiations. Using the Standard Material Transfer Agreements of the Pandemic Influenza Preparedness Framework, and principles from contract law, his team at the Global Health Centre has parsed out the level of detail countries will need now, and those that can be left for later.
This would be helpful in understanding what a PABS contract can look like. We are grateful to Strobeyko and colleagues, for making this analysis available to our readers. Find below a link to the analysis of publicly available agreements, provision by provision, using the PIP Framework template, and the most common language across signed agreements as baselines.
We hope you find this timely, as countries gather next week for a resumed session of the PABS negotiations in Geneva.
And yes, we are open to sponsorship on a case-by-case basis, on these pages. See below for more details.
Geneva Health Files offers value to our readers who are experts in global health. Tracking global health policy-making in Geneva is tough and expensive, without institutional support. For six years, we have provided you with the information and tools that directly contribute to your work in the field. We rely on our readers who value news they can use.
By becoming a paying subscriber you make our work possible. Our subscribers contribute towards greater accountability in global health.
More soon.
Priti
Priti Patnaik, Founder & Publisher, Geneva Health Files
Feel free to write to us: genevahealthfiles@gmail.com ; Find us on BlueSky, Instagram and Linkedin.

Striking a Balance: Standardization and Flexibility in Pandemic Contracts
What a comparison of PIP Framework benefit-sharing agreements shows for PABS negotiations
By Adam Strobeyko
Strobeyko is a Legal Advisor & Researcher at the Global Health Centre, Geneva Graduate Institute. He can be reached here: adam.strobejko@graduateinstitute.ch

The inclusion of contracts in the Pathogen Access and Benefit Sharing (PABS) System remains one of the most contentious issues in the ongoing negotiations of the Intergovernmental Working Group (IGWG) at the WHO.
I have previously compared the drafting of PABS contracts to recounting a winding love story to travelling companions, only to find the telling constantly derailed by their interruptions. After the previous meeting of IGWG6, though, a better parallel may be Laurence Sterne's Tristram Shandy, whose narrator sets out to write a complete account of his life, but finds that the more meticulously he tries to capture everything, the further behind he falls. By the third volume of this series, he is yet to be born!
Similarly, inclusion of contracts in the Pandemic Agreement and its PABS Annex has resurfaced throughout the negotiations, only to be overtaken by new complications. It is increasingly clear that the future PABS System will rely on "legally binding contracts" for benefit sharing. But how those contracts will actually work — their legal substance, any latitude for case-by-case negotiations with PABS material recipients, and their enforceability — remain an open question. An agreement on the core and basic tenets is still pending. In addition, negotiating detailed contract terms within the IGWG risks complicating already complex discussions.
Luckily, when negotiators sit down to design PABS contracts, they will not start from scratch. In this article, I situate the debate in light of lessons from the PIP Framework's use of Standard Material Transfer Agreements (SMTAs) and contract law principles. I draw on them as suggestions for the design of contracts governing pathogen exchange under the Pandemic Agreement and its Annex.
To this end, we have prepared a comparison table of signed SMTA2s. The table compares 17 publicly available Category A SMTA2s (Vaccine & Antiviral Manufacturers) provision by provision, using the PIP Framework template and the most common language across signed agreements as baselines. Cells are color coded to indicate use of standard language and to flag deviations, with verbatim contract text included where provisions diverge substantively from the baselines.
I argue that PABS contracts should be more standardized than PIP SMTAs on core legal issues such as enforcement, benefit-sharing, dispute settlement, and force majeure. But does that require negotiating every clause in advance and treating it as fixed? Not necessarily. A closer look at PIP SMTA2s reveals that strong standardization on core provisions has coexisted with flexibility.
A separate and an equally pertinent question, largely unexamined so far, is what happens when such contracts are embedded not in a WHA resolution, as with PIP, but in a treaty requiring ratification. I explore below the experiences of the Plant Treaty that needed domestic ratification of the international agreement that included contracts.
Examining PIP Framework SMTAs
Under the PIP Framework, pandemic influenza viruses are shared through WHO's Global Influenza Surveillance and Response System (GISRS), including onward transfers to recipients outside the network, such as pharmaceutical companies. In return, entities receiving PIP biological materials undertake to provide benefits linked to their use, channeled through WHO.
The PIP Framework is not a legally binding instrument. Rather, benefit-sharing commitments are formalized through private-law contracts known as SMTAs. SMTA2s are negotiated and concluded between WHO and companies. They generally do not preclude companies from seeking intellectual property rights on products, while requiring them to provide benefit-sharing commitments selected from an annexed menu (see model SMTA2 text in PIP Framework Annex 2). These may include donating or reserving shares of real-time pandemic production for WHO allocation at affordable prices, and/or granting licenses or transferring technology to support manufacturing capacity in developing countries.
To date, WHO has concluded 17 SMTA2s with Category A manufacturers (vaccine and antiviral producers), securing, at least on paper, over 940 million doses of future pandemic vaccines for allocation to countries in need.
Our comparison table lays these 17 agreements side by side, provision by provision. The patterns that emerge are revealing and directly relevant to the PABS design challenge.
A Standardized Core
A first observation is how much of the contract text is effectively identical across all 17 agreements. The subject matter, biosafety handling obligations, privileges and immunities, warranties, and dispute resolution clauses are reproduced in near-identical language from one signed SMTA2 to the next, with only minor stylistic variations.
That the signed agreements resemble each other may not seem surprising: these are, after all, meant to be standard. But the convergence is not simply a matter of copying the PIP Framework template. In fact, signed SMTA2s depart from or elaborate on the PIP template language, while converging among themselves, which hints at harmonization achieved by WHO while negotiating them with companies.
More striking still is what happened with provisions that the PIP Framework SMTA2 template left deliberately open. On dispute settlement, liability, duration, and termination, the template says only "to be agreed by the parties."
Yet all 17 signed agreements converged on the same solutions: dispute settlement including a three-month period of negotiation or other amicable settlement (including conciliation), failing which disputes proceed to binding arbitration under the rules of International Chamber of Commerce before a three-member panel seated in Geneva. Swiss law governs any question of interpretation or application not addressed by the SMTA2 terms.
A common contractual framework thus emerged organically through WHO's repeated bilateral negotiations. For PABS, this suggests that a degree of discretion may reasonably be afforded to WHO in developing contract provisions through its negotiations with companies, provided that core parameters are agreed in advance.
Flexibility Where it Counts
If the legal skeleton is uniform, the benefit-sharing commitments are where the real negotiation happened. The PIP model SMTA2 template offers a menu of benefit-sharing options, which include donating or reserving at affordable prices "at least 10%" pandemic vaccine production. The reality departs from the PIP Framework template, yet it is also surprisingly harmonized across signed contracts.
The most common structure — 8% donation and 2% reservation — was adopted by 11 of the 17 manufacturers. The splits vary among the rest: one committed to 5% and 5%, another to 7.5% and 7.5% (the highest combined commitment at 15%), another to 10% and 2.5%. These terms were clearly subject to case-by-case negotiation, even as the majority gravitated toward a common formula.
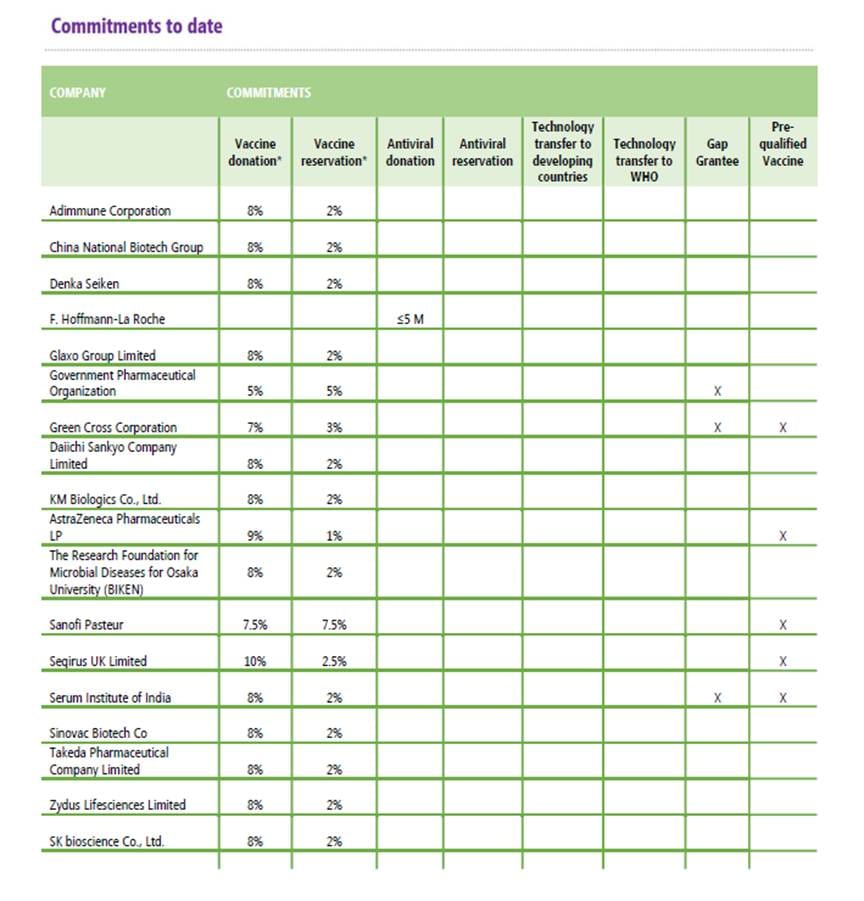
Source: WHO, 'SMTA2 Negotiating Strategy and Achievements: Vaccine Manufacturers At-A-Glance' (updated 30 March 2026), available at https://www.who.int/initiatives/pandemic-influenza-preparedness-framework/smta2-benefits.
Technology Transfer As An Option With No Uptake
Another pattern stands out starkly: not a single Category A manufacturer selected any of the technology transfer options listed in the model template: licensing to developing-country manufacturers or royalty-free licenses to WHO. These options existed on paper but turned out not to be included in contracts with the manufacturers.
Meanwhile, in the PABS negotiations, technology transfer and non-exclusive licenses are key demands for some countries.
Shipment Notice As A Pragmatic Workaround
The PIP Framework also produced an instructive workaround when the contractual system proved too rigid for operational reality. Under the Framework, PIP biological materials may only be shared with a non-GISRS entity that has already signed an SMTA2 with WHO. In practice, this precondition risked paralyzing timely virus sharing. WHO and its partners therefore developed an interim process under which a "Shipment Notice" alerts the recipient that the requested materials carry PIP Framework obligations, and shipments proceed while SMTA2 negotiations continue. If the recipient does not accept those obligations, it must cancel its request.
The Dangers Of Confidentiality: Red Flags on Term Sheets
The PIP contracts also have term sheets, which are confidential and annexed to each agreement, contain the actual operative details on liability, indemnity, and operational parameters.
The most commercially sensitive provisions sit behind a veil of confidentiality that makes independent scrutiny difficult.
The problem runs deeper than mere opacity. Rourke's textual analysis of the PIP Framework's SMTA2s shows that liability, indemnity, and force majeure definitions — some of the most consequential provisions for pandemic contracts — are precisely the ones relegated to confidential term sheets. Her analysis of the only publicly available force majeure provision from old SMTA2 between WHO and Glaxo Group Limited on December 18, 2012 shows it listing "disease (including influenza pandemic)" among the events relieving a manufacturer of its delivery obligations, thus undermining the very obligations the contract exists to secure. Whether the remaining term sheets contain similarly self-defeating language is unknowable from the outside.
This provides to serve as a cautionary note for PABS negotiators.
Lessons For Standardization of PABS Legally Binding Contracts
What does all this mean for PABS? The PIP data yields three clear lessons.
First, legal standardization can work organically when it is carefully tempered. In the context of PIP SMTA2s it was not a product of a single drafting exercise but of a living process: WHO negotiating contract after contract with manufacturers and, over time, arriving at common solutions. Threading a delicate line between a priori standardization and flexibility is necessary if the system is to deliver both legal clarity and be attractive to companies.
Second, benefit-sharing needs structured flexibility. The PIP experience shows that the headline benefit-sharing figures (“at least 10%”) masked significant variation in practice, and that options requiring deeper commitments around technology transfer were systematically avoided. PABS should thus define minimum floors and mandatory commitment categories. The menu of benefit-sharing options could be narrower but mandatory, with flexibility potentially allowed in how manufacturers meet each floor, and not in whether they do so at all. It is perhaps better to build in a degree of flexibility from the start than to improvise when the system proves too rigid in operational reality.
Third, transparency is a precondition for accountability. The confidential term-sheet model adopted under PIP makes independent assessment of compliance nearly impossible. Force majeure should be addressed transparently in the main contract text, through an exhaustive list of qualifying events and clear procedural requirements. A “pandemic emergency,” as defined by the Pandemic Agreement, must not be interpretable as a force majeure event, as doing so would defeat the very object and purpose of the Agreement.
Contracts Within a Ratified Treaty: A New Dynamic?
There is a further dimension the PABS debate has not yet fully reckoned with. The PIP Framework is a non-binding instrument adopted by the WHA under Article 23 of the WHO Constitution and never had to pass through national ratification. The Pandemic Agreement, negotiated under Article 19 of the Constitution, is a treaty. If its PABS Annex includes standardized contracts between WHO and private manufacturers, its success will depend on the success of both legal instruments taken together.
Treaties with contractual mechanisms almost always require domestic implementing legislation, and embedding contract templates in a treaty annex puts commercial terms in front of parliamentarians who would not normally deal with them, giving them ample material for objection or for requiring governments to file reservations to those terms upon ratification of the treaties. It should be noted, in this connection, that Article 25 of the Pandemic Agreement allows for reservations not incompatible with the object and purpose of the Agreement.
The Plant Treaty, the closest analogue, mandates an SMTA for exchanges of plant genetic material. Tvedt's analysis showed why Plant Treaty SMTAs never delivered meaningful monetary benefit-sharing. Their core terms were products of diplomatic compromise rather than legal drafting, leaving interpreters unable to determine when exactly a payment obligation is triggered. With no remedies specified for breach and the FAO designated as enforcer but lacking any institutional incentive to sue private corporations, the instrument looked like a contract but could not function as one.
The US track record with comparable treaties deserves attention. It signed the Plant Treaty in 2002 but never ratified it, with congressional concerns centering on intellectual property implications and future financial commitments. Its decades-long refusal to ratify UNCLOS turns in significant part on Senate objections to the International Seabed Authority, a body empowered by the treaty to enter contracts directly with private mining companies.
Whether the Pandemic Agreement's contractual architecture will face similar headwinds in other countries is an open question, but it is one that PABS designers would do well to consider.
This calls for a two-step approach: the treaty sets out the mandate, core principles, and minimum parameters, while the detailed contract template gets adopted later by the COP, as the Plant Treaty did with its SMTA. Thus, the template can evolve without reopening the treaty, which reflects how PIP's own SMTA2 provisions have shifted across successive rounds of negotiations.
Conclusions
Tristram Shandy does eventually get born — it just takes longer than anyone expected. The PABS contract debate may follow a similar arc. The PIP experience shows that standardization is not only feasible but already proven in practice, and that flexibility on benefit-sharing modalities can coexist with a firm legal core. It also suggests that sensitive provisions would gain legitimacy if embedded in a publicly available standard agreement rather than buried in confidential term sheets.
The ratification dimension reinforces the case for separating the political question from the technical one.
A treaty text that establishes the mandate for standardized contracts, sets out core principles, and defines minimum benefit-sharing floors gives parliaments something concrete to approve without requiring them to parse commercial fine print. The detailed contract template can then be developed by the COP, drawing on technical and legal expertise.
Getting this balance right matters. A system that defers these questions indefinitely or buries them in vague language may look comprehensive on paper but will struggle to deliver when a pandemic actually arrives. The PIP Framework, for all its imperfections, demonstrates that workable contracts between an international organization and private companies can be built, refined, and sustained over time. PABS has the advantage of learning from that experience. And after facing many challenges, even Tristram Shandy got there in the end.
We thank colleagues from the Governing Pandemics initiative at the Global Health Centre, Geneva Graduate Institute: Suerie Moon, Gian Luca Burci, and Daniela Morich for thorough review and comments on earlier drafts of this piece.
This article expands on an earlier piece published in the Governing Pandemics Snapshot, a series of briefings on global reforms for pandemic preparedness and response produced by the Global Health Centre at the Geneva Graduate Institute. The series is available here: [Link].
Other relevant readings on this topic:






SPONSORSHIP OPPORTUNITIES IN OUR NEWSLETTER
Want visibility in Global Health? Our readership includes diplomats, policymakers, experts, the private sector, academia, civil society organizations, and students. Consider becoming a sponsor!

Global health is everybody’s business. Help us probe the dynamics where science and politics interface with interests. Support investigative global health journalism.







![Participation is The Medicine For AI Healthcare, But It Needs To Be Real [Guest Essay]](https://storage.ghost.io/c/3e/27/3e275aa8-3271-4edb-ac7b-04dcacf8a053/content/images/size/w30/2026/06/pexels-steve-13551570.jpg)

